 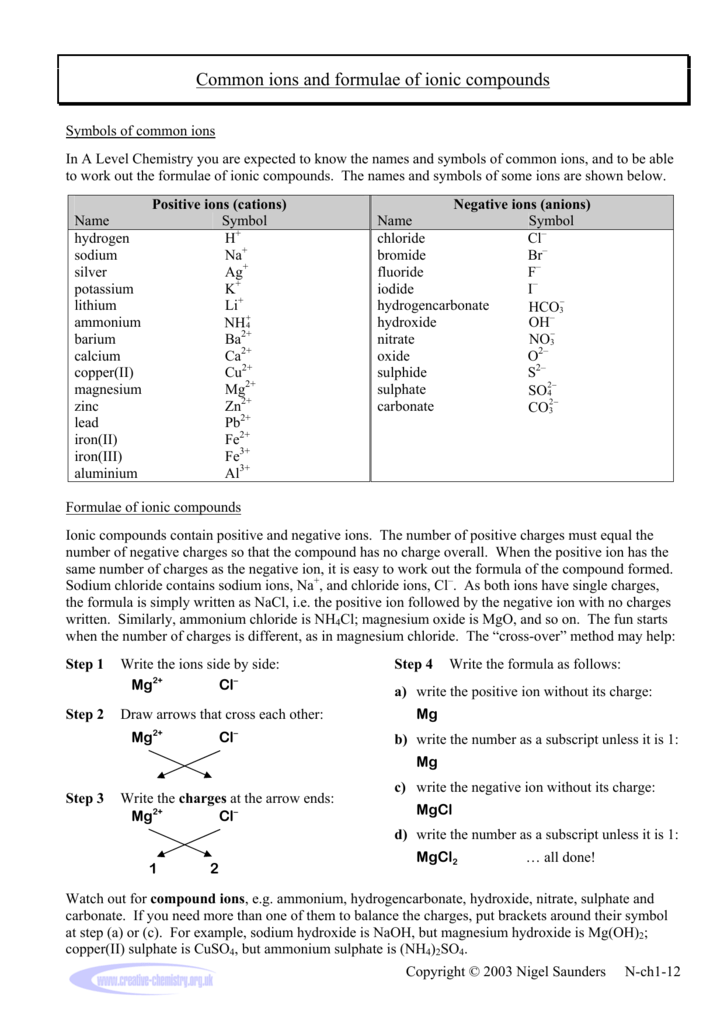
The formula for an ionic compound is called an formula. Crystals are 3 dimensional arrays of positive and negative ions in a pattern. Instead of forming molecules, ionic compounds form. Solid sodium chloride contains equal numbers of cations (Na +) and anions (Cl −), thus maintaining electrical neutrality. Another name for an ionic compound is an. Three characteristics of ionic compounds you might have listed are that they are electrically neutral, dissolve only in polar solvents, and, as solids, are hard and brittle.\). Common table salt ( N a C l NaCl N a Cl) forms this way, but so do ionic lattices such as:Ĭopper (II) sulfate ( C u S O 4 ( H 2 O ) x CuS N a H C O 3 (baking soda). Ternary ionic compounds always contain three elements in their formula, including a positive ion (cation) and a negative ion (anion). Ionic compounds form crystalline structures called crystal lattices. When one element may be a metal and therefore the other may be a non-metal, then two-element compounds are usually ionic compounds. The compound is neutral overall, but consists of positively charged ions called cations and negatively charged ions called anions. Ions with net positive charges are called cations, pronounced “cat-ions.” Metals are commonly cations. Ions that are negatively charged are called anions, pronounced “an-ions.” Common anions are non-metals. 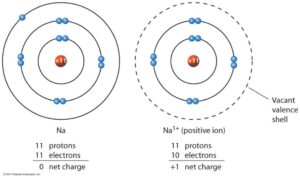
A good example is the ammonium ion made up of one nitrogen atom and four hydrogen atoms. One example of an ionic compound is sodium chloride (NaCl Figure 2.7.3 ), formed from sodium and chlorine. Polyatomic ions are ions comprised of more than one atom. If the electrostatic energy is positive, the particles repel each other if the electrostatic energy is negative, the particles are attracted to each other. Positively charged ions are called cations negatively charged ions, anions. Two-element ionic compounds and polyatomic ionic compounds are equally common. Key People: ion, any atom or group of atoms that bears one or more positive or negative electrical charges. Google Classroom Learn how to name monatomic ions and ionic compounds containing monatomic ions, predict charges for monatomic ions, and understand formulas. Ionic compounds are electrically neutral and can form from just a few atoms (ions) or from large numbers of atoms (ions). Most atoms join by covalent bonding, in which shared electrons form directional bonds. Ionic compounds are pure substances formed from chemically bonded ions. Atoms lose and gain electrons as they react to become more stable. The number of electrons lost or gained determines the ion's charge. The water molecules can pull hard enough to eventually break each salt molecule. When the salt is put in water, the water is pulling Na+ on one side and Cl- on the other side. Also, ionic compound tend to form complex lattice networks and structures (see the picture). Ion – An atom that has gained or lost an electron, creating a charged particle that is either positive (more protons than electrons) or negative (more electrons than protons) Water is a covalent polar compound (it has positive and negative poles). Nucleus – The center of every atom, consisting of positively charged protons and neutral neutronsĮlectron – The tiny, negatively charged particle orbiting the nucleus in the electron cloud and held to the atom by the positive-negative attraction between it and the nucleus’s proton Let’s review some basics of atomic structure: To fully understand ionic compounds, we need to understand what ionic compounds are composed of. Individual atoms can gain or lose electrons. 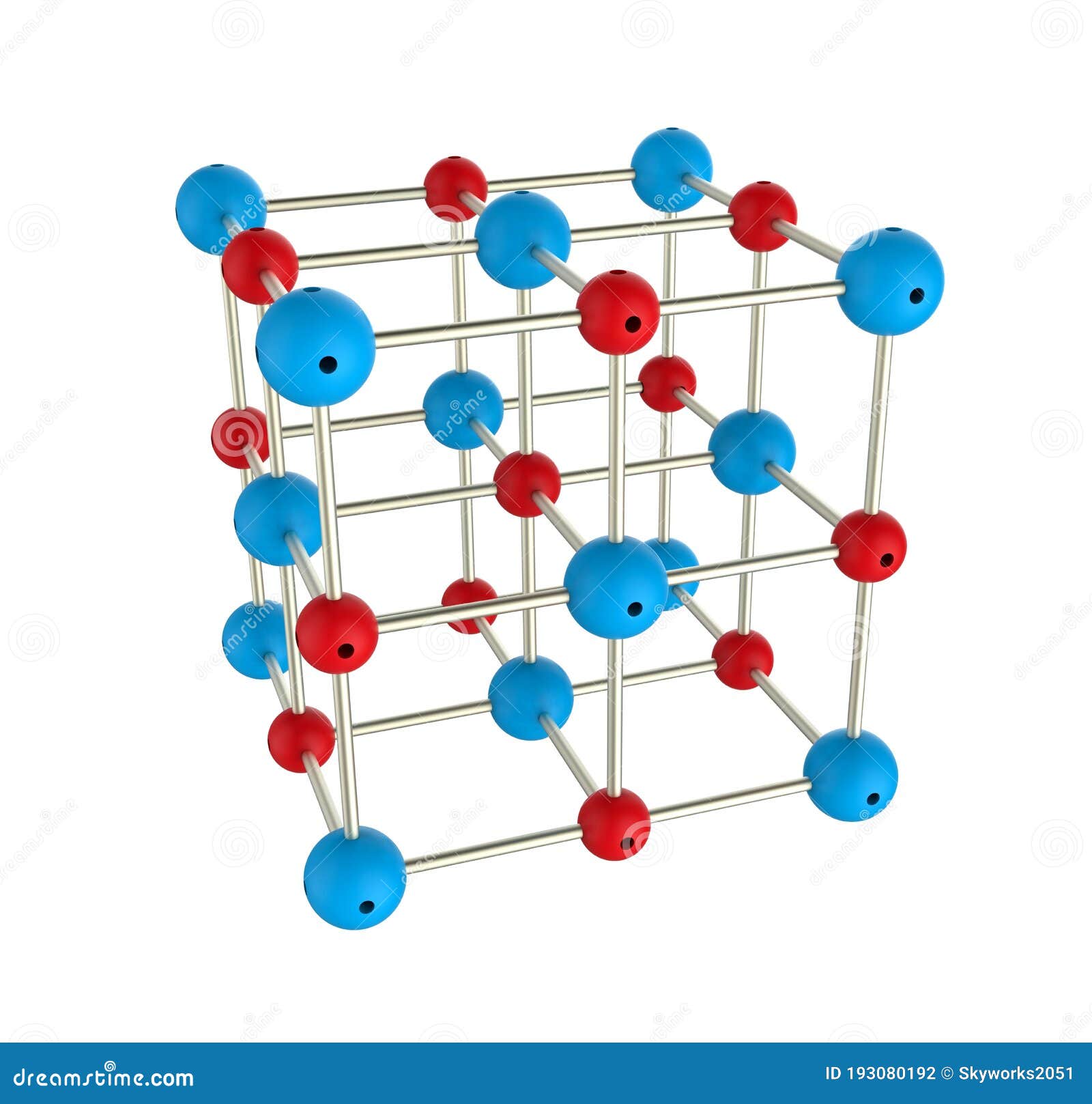
What is an ionic compound What are ionic compounds made of? Compounds formed from positive and negative ions are called ionic compounds. Some minor electron sharing always occurs, but the compound is chiefly formed by ions. When writing the Ls symbol is written first, and the Ls symbol. There could be elemental ions, atoms that have lost or gain electrons. Ionic compounds are pure substances formed primarily by positive and negative ion attraction. Transcribed Image Text:Part 2: Ternary Compounds Ternary ionic compounds always contain a ion. The naming of ionic compounds that contain polyatomic ions follows the same rules as the naming for other ionic compounds: simply combine the name of the cation and the name of the anion. Ionic compounds are compounds that are composed of discrete ions or charged species. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
